research interest
The bloodfeeding interface and pathogen transmission
Our work over the past 27 years has collectively provided novel insights into the factors in mammalian blood that can function inside the mosquito host following ingestion.
Mosquitoes ingest blood to produce eggs, but many factors present in blood, including peptides, proteins, vitamins, small molecules, biogenic amines, drugs, hormones and pathogen-derived factors activate highly conserved and ancient signaling pathways in multiple mosquito tissues to control lifespan, behavior, reproduction, and susceptibility to infection.
In this context, blood is both food and information to the mosquito host, controlling the likelihood that pathogen transmission will occur. Studying these phenomena has enabled us to characterize a variety of ancient cell signalling pathways in the mosquito host that are new to our understanding of innate immunity in several important mosquito vector species.
In this context, we can leverage the complexity and opportunity of the bloodfeeding interface to design novel control strategies for mosquito vectors of malaria parasites and arboviruses.
research interest
Malaria-induced allergic inflammation: the gut barrier and parasite transmission
Parasitic infections are often associated with the development of an array of symptoms consistent with allergy, including activation of basophils, mast cells and eosinophils as well as the generation of histamine, alterations in host metabolism and skewing of the host immune response to T helper type 2 or (Th2) cellular and humoral factors.
Published studies have shown that malaria induces allergic inflammation, but the effects of this inflammation and its functions in the host response to infection and in parasite development have not been clear.
Our studies have demonstrated that malaria-induced “leaky gut” and the translocation of enteric bacteria into the blood (bacteremia) result from intestinal mast cell influx and activation. A Th2-biased response to infection precedes this mast cell activation and is characterized by the early appearance of IL-4, IL-10, mast cell protease (Mcpt)1 and Mcpt4, and early increases in circulating basophils and eosinophils.
Quite unexpectedly, we have discovered that this Th2-biased response not only controls intestinal barrier integrity, but also parasite transmission to Anopheles mosquitoes.
We are currently exploring the role of early cytokines in the activation of basophils and mast cells and how basophil- and mast cell-derived factors control host responses in the intestine, the spleen and in circulation to coordinate these diverse parameters of infection.
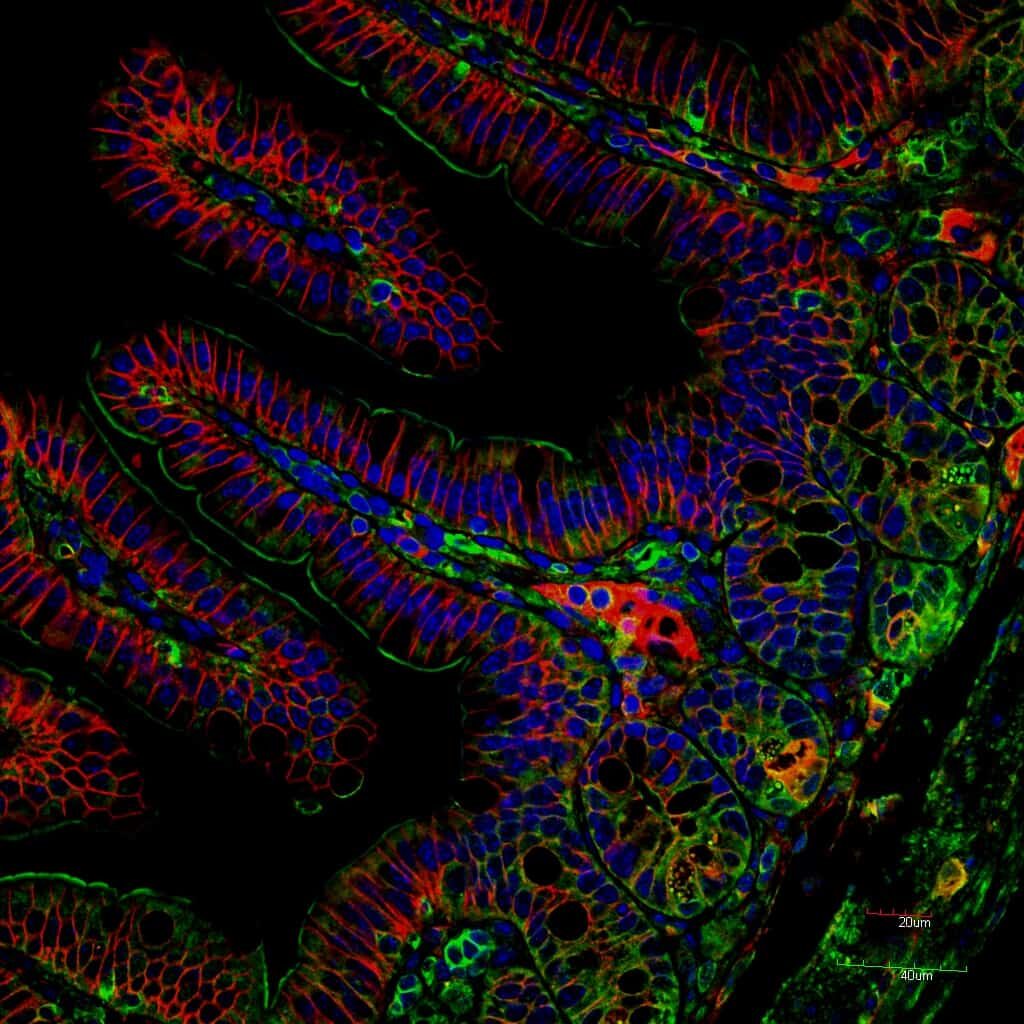
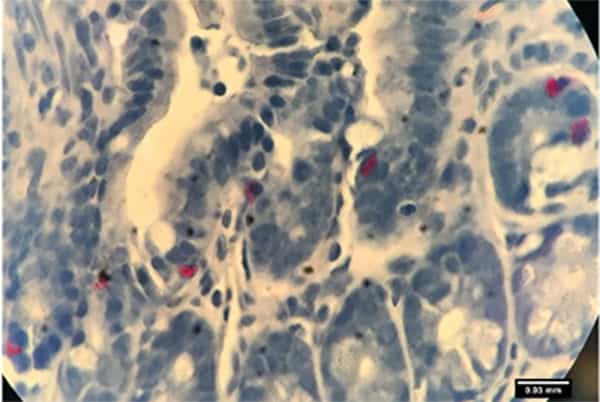
The Gut Barrier
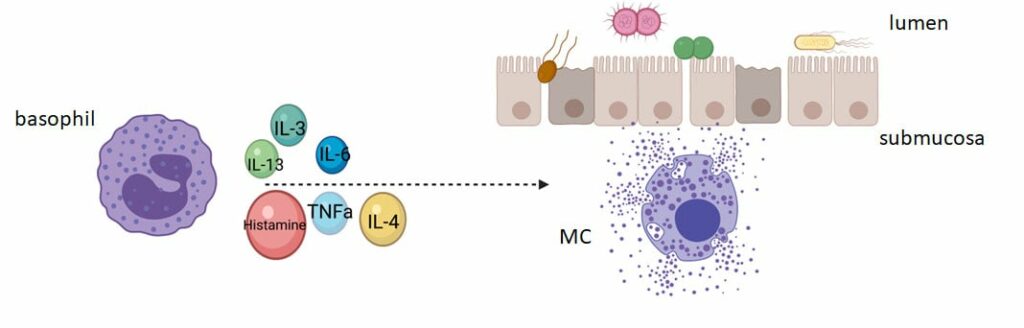
Data from our NIH-funded studies suggest that activated basophils and mast cells, shown here, contribute to malaria-induced degradation of the intestinal barrier or “leaky gut”, resulting in the translocation of enteric bacteria into the circulation. Malaria-associated bacteremia has been reported in both children and adults with malaria and these cases tend to have more severe symptoms and higher case fatality rates compared to individuals without bacteremia (reviewed in Donnelly et al. 2021 Curr Res Microb Sci 2:100036 doi: 10.1016/j.crmicr.2021.100036).